 Use the buttons above to change your view of the periodic table and view Murray Robertson’s stunning Visual Elements artwork. Click the tabs at the top to explore each section. Figure 2 2: Within each period, the trend in atomic radius decreases as Z increases for example, from K to Kr. The Royal Society of Chemistrys interactive periodic table features history, alchemy, podcasts, videos, and data trends across the periodic table. For example,let us consider the nature of the second group hydroxides:īe(OH) 2 amphoteric Mg(OH) 2 weakly basic Ba(OH) 2 strongly basic Beryllium hydroxide reacts with both acid and base as it is amphoteric in nature.\). The stronger pull (higher effective nuclear charge) experienced by electrons on the right side of the periodic table draws them closer to the nucleus, making the covalent radii smaller. Hence, the hydroxides of these elements become more basic. Know everything about Neon Facts, Physical Properties, Chemical Properties, Electronic configuration. 
It is part of group 18 (helium family or neon family). Neon, symbol Ne, has a Face Centered Cubic structure and Colorless color. Thus, the elements from the two extreme ends of the periodic table behave differently as expected.Īs we move down the group, the ionisation energy decreases and the electropositive character of elements increases. Element 10 of Periodic table is Neon with atomic number 10, atomic weight 20.1797. 
Conversely Cl 2O 7 gives strong acid called perchloric acid upon reaction with water So, it is an acidic oxide. Since sodium oxide reacts with water to give strong base sodium hydroxide, it is a basic oxide. 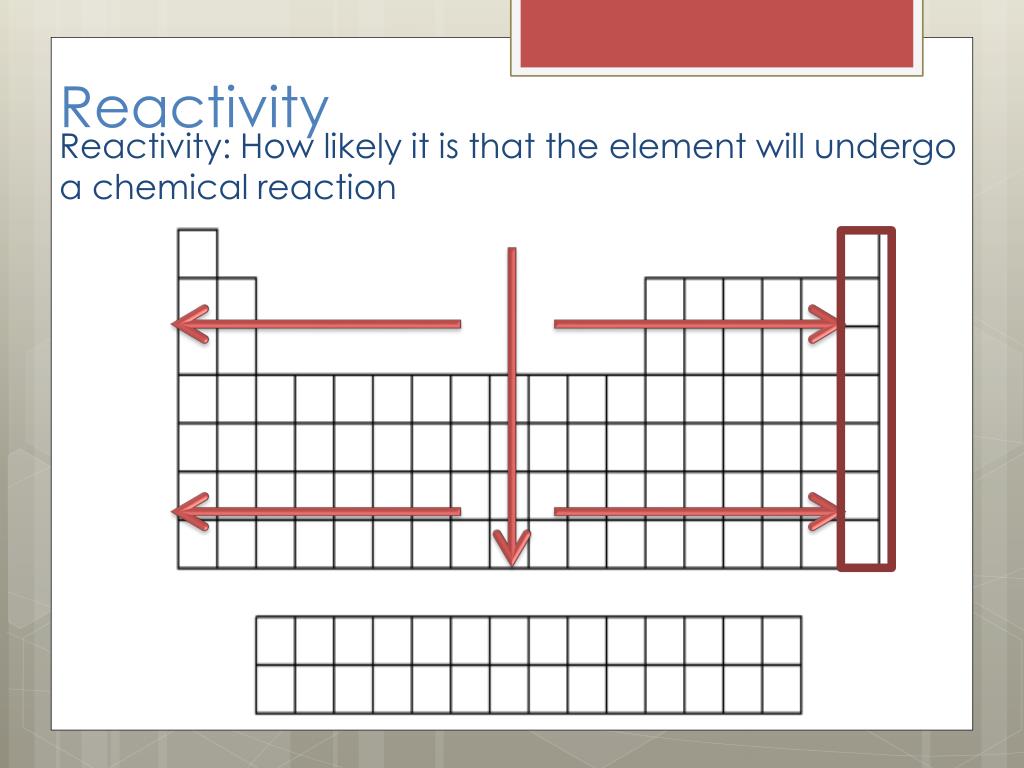
Consider the reaction of alkali metals and halogens with oxygen to give the corresponding oxides. It can be used as a differentiated activity for the more able students within a group. This could be used to follow up some work on the periodic table where the trends in reactivity in groups 1 and 7 have been identified. Let us analyse the nature of the compounds formed by elements from both sides of the periodic table. Chemistry for the gifted and talented: trends in reactivity in the periodic table. On the other hand the elements located in the top right portion have very high ionisation energy and are non-metallic in nature. The ionisation energy is directly related to the metallic character and the elements located in the lower left portion of the periodic table have less ionisation energy and therefore show metallic character. Because nonmetals have exactly the opposite property than that of the metals. Learn how metals and halogens vary in reactivity across the periodic table with this short series by Stile Education. The noble gases having completely filled electronic configuration neither accept nor lose their electron readily and hence they are chemically inert in nature. Reactivity is a measure of how easily an element will combine with other elements to form compounds. As a consequence of this, elements of these extreme ends show high reactivity when compared to the elements present in the middle. Learn about the trends in periodic table : Get detailed article on periodic table trends, overview, physical properties, chemical properties and examples. On the other hand, the elements on right side of the periodic table have high electron affinity and readily accept electrons. The elements on the left side of the periodic table have less ionisation energy and readily loose their valence electrons. 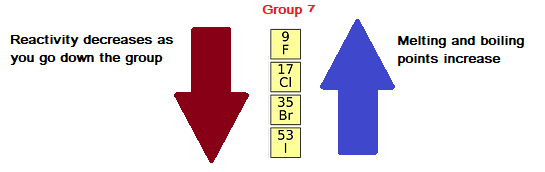
The physical and chemical properties of elements depend on the valence shell electronic configuration as discussed earlier. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
